| ON THE COVER | 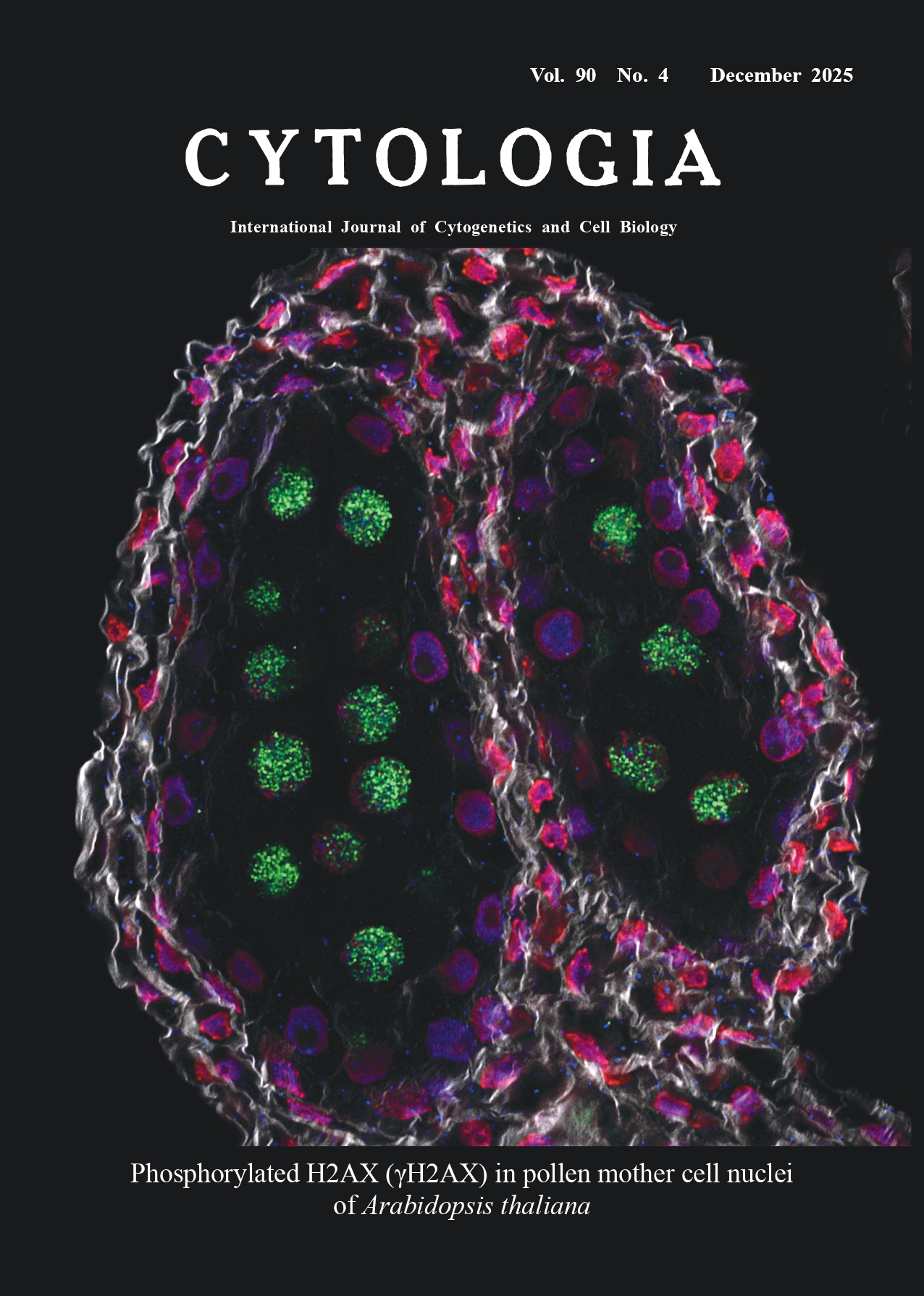 |
||
|---|---|---|---|
| Vol. 90 No.4 December 2025 | |||
| Technical Note | |||
|
|
|||
| In situ detection of γH2AX in developing Arabidopsis anthers by whole-mount immunofluorescent
chemistry
Keiichi Takagi* The Wakasa Wan Energy Research Center, 64–52–1 Nagatani, Tsuruga, Fukui 914–0192, Japan
Histone H2AX is one of histone H2A subtypes. When DNA double-strand breaks (DSBs) are generated by genotoxic stress, DNA damage response (DDR)-related PIK3-like protein kinases convert H2AXs along break points into the phosphorylated form of H2AX (γH2AX). This reaction is one of the earliest events of the DDR and is often used as a marker of DSB formation in cells. Meiotic recombination is initiated by spontaneous DSB formation induced by SPO11(s). Phosphorylation of H2AX also occurs after such spontaneous DSB formation. In Arabidopsis, it has been reported that γH2AX was detected in nuclei of pollen mother cells (PMCs) between late G2 and meiotic prophase I (Sanchez-Moran et al. 2007). The cover image is an optical section of an anther of Arabidopsis thaliana at stage 9 of flower development (Alvarez-Buylla et al. 2010), showing γH2AX in PMC nuclei. The staining method is as follows. After fixation and removal of pigments and lipids, hydrogel solution was infused and polymerized in the flower buds according to the method described by Chung et al. (2013). The tissues were then subjected to whole-mount immunocytochemical techniques described by Sauer et al. (2006) with various modifications. Briefly, the cell walls of the tissues were digested in an enzyme cocktail containing cell wall-digesting enzymes and DNase inhibitors at 4°C for 7 d. After the digestion, the tissues were incubated with primary antibodies at 4°C for 7 d. As the primary antibodies, a mouse monoclonal antibody raised against the γH2AX C-terminal oligopeptide (NKGDIGSApSQEF) and a rabbit polyclonal antibody raised against the unphosphorylated form were simultaneously applied to detect γH2AX and H2AX, respectively. After washing with PBST, the tissues were incubated with secondary antibodies using anti-mouse IgG AlexaFluor488 and anti-rabbit IgG AlexaFluor568 at 4°C for 7 d. Nuclei were co-stained with NucSpot650/665 during the incubation. After the incubation, undigested cell wall was stained with Calcofluor White M2R and whole tissues were mounted using ProLong Glass antifade mountant (Thermo Fisher, United States). The Airyscan imaging of LSM900 (Carl Zeiss, Germany) was used to acquire optical sectional images of each fluorescence, and a merged fluorescent image was created by ImageJ. In the cover image, anti-γH2AX binding is shown in green, anti-H2AX binding in blue, the nucleus in red, and the cell wall in grey, respectively. Strong fluorescence of anti-γH2AX binding is detected in nuclei of PMCs inside an anther. Nuclear morphology of PMCs revealed they were in leptotene stage (not obvious in the figure). Although other nuclei show the anti-H2AX binding, there is no apparent anti-γH2AX signal in them, suggesting that DSBs were specifically induced in PMCs. Squashed tissue preparation, that has been used in most of the previous plant γH2AX research, resulted in the loss of spatial information. The whole-mount preparation in the present note may be applicable for elucidating regional differences in the γH2AX response of plants after exposed to genotoxic stress.
Chung, K., Wallace, J., Kim, S. Y., Kalyanasundaram, S., Andalman, A. S., Davidson, T. J., Mirzabekov, J. J., Zalocusky, K. A., Mattis, J., Denisin, A. K., Pak, S., Bernstein, H., Ramakrishnan, C., Grosenick, L., Gradinaru, V., and Deisseroth, K. 2013. Structural and molecular interrogation of intact biological systems. Nature 497: 332–337. Sanchez-Moran, E., Santos, J.-L., Jones, G. H., and Franklin, F. C. H. 2007. ASY1 mediates AtDMC1-dependent interhomolog recombination during meiosis in Arabidopsis. Genes Dev. 21: 2220–2233. Sauer, M., Paciorek, T., Benková, E., and Friml, J. 2006. Immunocytochemical techniques for whole-mount in situ protein localization in plants. Nat. Protoc. 1: 98–103. * Corresponding author, e-mail: ktakagi@werc.or.jp DOI: 10.1508/cytologia.90.201 |
|||